Blog
Rod-Cone Dystrophy and Macular Dystrophy
March 29, 2026
Introduction
Rod-Cone Dystrophy (RCD) and Macular Dystrophy (MD) are part of a broader category known as inherited retinal diseases (IRDs) conditions characterized by progressive degeneration of photoreceptor cells in the retina. These photoreceptors "rods and cones" are essential for converting light into neural signals, enabling vision. As these cells deteriorate, patients experience gradual vision loss that may ultimately lead to blindness.
Despite major advances in genetics and molecular ophthalmology, conventional treatments remain limited. There is currently no widely available cure, and most interventions aim to slow progression rather than restore vision.
Emerging therapies such as gene therapy and stem cell therapy show promise but are restricted to specific mutations or remain experimental.
This therapeutic gap is where Netra Restoration Therapy (NRT) positions itself—not as a replacement for modern ophthalmology, but as a complementary, systems-based approach targeting recovery-limiting biological dysfunctions that contribute to retinal degeneration.
Understanding the Disease Process
Rod-Cone Dystrophy
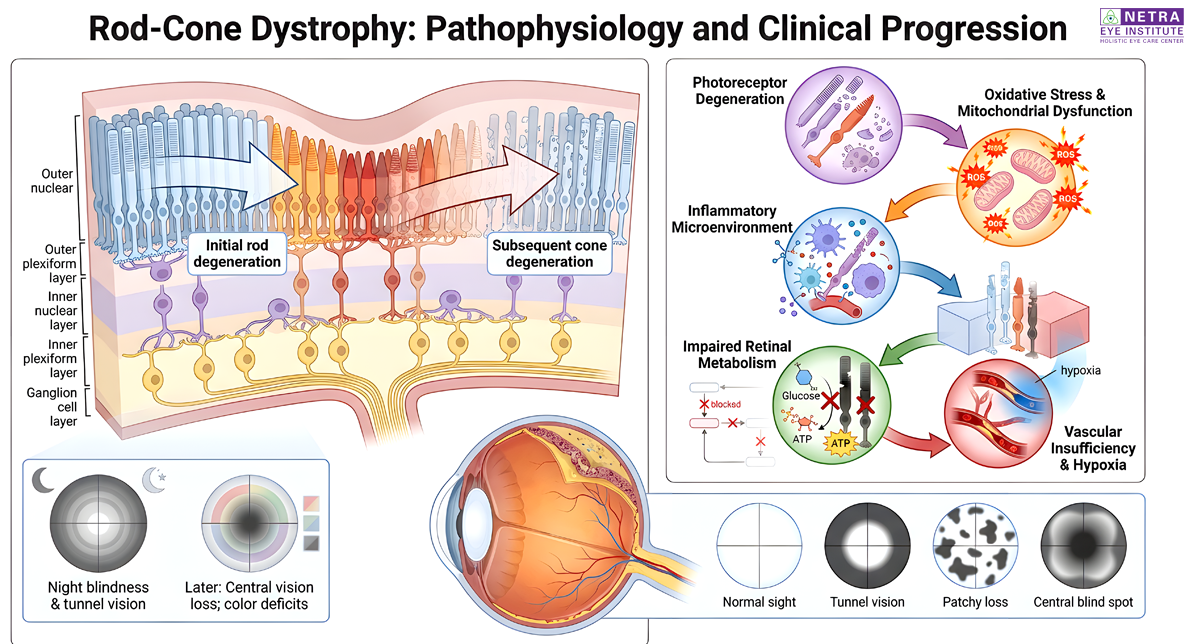
Rod-Cone Dystrophy typically begins with degeneration of rod photoreceptors, affecting night and peripheral vision, followed by progressive cone loss, impacting central and color vision. Over time, the disease leads to widespread retinal dysfunction and visual disability.
Macular Dystrophy
Macular Dystrophy primarily affects the macula—the central region of the retina responsible for detailed vision. Patients experience progressive central vision loss, affecting reading, facial recognition, and fine visual tasks.
Shared Pathophysiology
Both conditions share several underlying mechanisms:
- Photoreceptor degeneration (rods and cones)
- Oxidative stress and mitochondrial dysfunction
- Inflammatory microenvironment
- Impaired retinal metabolism
- Vascular insufficiency and hypoxia
Importantly, modern research highlights that preserving cone function is critical for maintaining central vision, even when rod degeneration begins first.
Limitations of Conventional Care
Current management strategies include:
- Vitamin supplementation
- Low vision aids
- Genetic counseling
- Experimental gene or cell therapies
However, these approaches largely:
- Focus on disease identification and monitoring
- Offer mutation-specific solutions
- Do not comprehensively address systemic and microenvironmental dysfunctions
As a result, many patients continue to experience progressive decline despite optimal conventional care.
The Netra Restoration Therapy (NRT) Approach
Netra Restoration Therapy introduces a multi-dimensional therapeutic model that addresses not just the retinal structure but the functional ecosystem of the eye.
Rather than targeting a single gene or pathway, NRT focuses on restoring the biological environment necessary for photoreceptor survival and function.
Core Principle:
Vision loss in RCD and MD is not only due to genetic damage—but also due to modifiable dysfunctions that limit recovery and regeneration.
Key Recovery-Limiting Biological Dysfunctions Addressed by NRT
1. Microvascular Insufficiency
Reduced retinal blood flow leads to chronic hypoxia, accelerating photoreceptor death. NRT aims to:
- Improve retinal perfusion
- Enhance oxygen and nutrient delivery
- Support retinal metabolic demand
2. Oxidative Stress
Photoreceptors are highly metabolically active and vulnerable to oxidative damage. Research shows oxidative stress contributes significantly to retinal degeneration.
NRT addresses this through:
- Antioxidant modulation
- Cellular detoxification support
- Redox balance restoration
3. Mitochondrial Dysfunction
Photoreceptor cells rely heavily on mitochondrial energy production. Dysfunction leads to:
- Reduced ATP generation
- Accelerated cell apoptosis
NRT strategies aim to:
- Enhance mitochondrial efficiency
- Improve cellular energy metabolism
4. Neuroinflammation
Chronic low-grade inflammation in the retina contributes to progressive degeneration.
NRT works to:
- Modulate inflammatory signaling
- Reduce microglial activation
- Promote a neuroprotective environment
5. Impaired Retinal Metabolism
Emerging therapies (such as RdCVF-based approaches) highlight the importance of metabolic support in preserving cone function.
NRT similarly emphasizes:
- Glucose metabolism optimization
- Nutrient delivery to photoreceptors
- Restoration of retinal biochemical pathways
How NRT Supports Photoreceptor Survival
1. Slowing Degeneration
By improving the retinal environment, NRT helps:
- Reduce oxidative and inflammatory stress
- Delay photoreceptor apoptosis
- Stabilize disease progression
2. Preserving Residual Vision
Even in advanced disease, some photoreceptors remain functional. NRT aims to:
- Enhance function of surviving rods and cones
- Improve visual performance (contrast, clarity, adaptation)
- Support neural signaling pathways
3. Enhancing Functional Vision
Patients often report improvements in:
- Night vision (rod function support)
- Central clarity (cone function support)
- Visual endurance and reduced fatigue
This aligns with research emphasizing functional endpoints such as mobility and light sensitivity in retinal disease trials.
4. Supporting Adjunctive Therapies
NRT can potentially complement emerging treatments such as:
- Gene therapy
- Stem cell therapy
- Neuroprotective pharmacology
By optimizing the retinal microenvironment, NRT may enhance responsiveness to these interventions.
Clinical Perspective
NRT represents a systems-biology approach to retinal disease:
- Moves beyond gene-centric treatment
- Addresses multi-factorial degeneration pathways
- Integrates metabolic, vascular, and neuroprotective strategies
- Providing a non-invasive, personalized therapeutic pathway
- Focusing on functional vision improvement, not just disease labeling
It aligns with evolving research trends emphasizing:
- Rods and Cones preservation
- Metabolic rescue
- Microenvironmental modulation